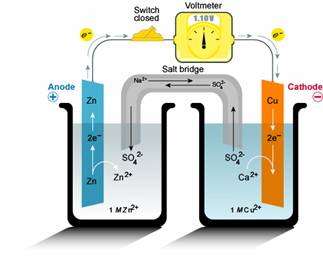
The basis for a battery operation is the exchange of electrons between two chemical reactions, an oxidation reaction and a reduction reaction. The key aspect of a battery which differentiates it from other oxidation/reduction reactions (such as rusting processes, etc) is that the oxidation and reduction reaction are physically separated. When the reactions are physically separated, a load can be inserted between the two reactions. The electrochemical potential difference between the two batteries corresponds to the voltage of the battery which drives the load, and the exchange of electrons between the two reactions corresponds to the current that passes through the load. The components of a battery, which are shown in the figure below, and consist of an electrode and electrolyte for both the reduction and oxidation reaction, a means to transfer electrons between the reduction and oxidation reaction (usually this is accomplished by a wire connected to each electrode) and a means to exchange charged ions between the two reactions.
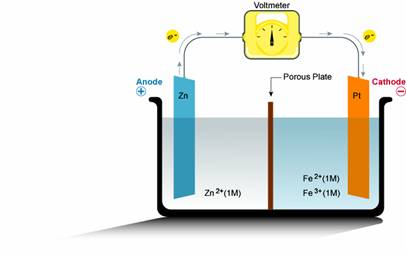
The key components which determines many of the basic properties of the battery are the materials used for the electrode and electrolyte for both the oxidation and reduction reactions. The electrode is the physical location where the core of the redox reaction – the transfer of electrons – takes place. In many battery systems, including lead acid and alkaline batteries, the electrode is not only where the electron transfer takes places, but is also a component in the chemical reaction that either uses or produces the electron. However, in other battery systems (such as fuel cells) the electrode material is itself inert and is only the site for the electron transfer from one reactant to another. For a discharging battery, the electrode at which the oxidation reaction occurs is called the anode and by definition has a positive voltage, and the electrode at which the reduction reaction occurs is the cathode and is at a negative voltage.
The electrode alone is not sufficient for a redox reaction to take place, since a redox reaction involves the interaction of more than a single component. The other chemical components of the reaction are contained in the electrolyte. For many practical battery systems, the electrolyte is an aqueous solution. One reasons for having an aqueous solution is the oxidized or reduced form of the electrode exists in an aqueous solution. Further, it is important that the chemical species in the electrolyte be mobile in order that they can move to the site on the electrode where the chemical reaction takes places, and also such that ion species can travel from one electrode to the other.
The current in the battery arises from the transfer of electrons from one electrode to the other. During discharging, the oxidation reaction at the anode generates electrons and reduction reaction at the cathode uses these electrons, and therefore during discharging, electrons flow from the anode to the cathode. The electrons generated or used in the redox reaction can easily be transported between the electrodes via a conventional electrical connection, such as a wire attached to the anode and cathode. However, unlike a conventional electrical circuit, electrons are not the only charge carrier in the circuit. Electrons travel from the anode to the cathode, but do not return from the cathode to the anode. Instead, electrical neutrality is maintained by the movement of ions in the electrolyte. If each redox reaction has a different electrolyte, a salt bridge joins the two electrolyte solutions. The direction of the ion movement acts to prevent a charge build-up at either the anode or the cathode. In most practical battery systems, the same electrolyte is used for both the anode and the cathode, and ion transport can take place via the electrolyte itself, eliminating the need for a salt bridge. However, in this case a separator is also inserted between the anode and the cathode. The separator prevents the anode and cathode from physically touching each other since they are usually in very close physical proximity to one another, and if they were to touch it would short out the battery as the electrons can be transferred directly without flowing through the external circuit and load.
The redox reactions which comprise a particular battery system define many fundamental parameters about the battery system. Other key battery properties, including as battery capacity, charging/discharging performance and other practical considerations are also influenced by the physical configuration of the battery, for example the amount of material in the battery or the geometry of the electrodes. The following pages describe how battery characteristics – voltage behavior, battery efficiency, battery non-idealities (self-discharge, degradation of battery capacity, etc) – are dependent on the operation of the redox reactions and the battery configuration.
